
The following symbols will be used in calculation formulas (units in square brackets): n Quantities are usually expressed in terms of M −1cm −1 or Lmol −1cm −1. The SI unit of molar attenuation coefficient is the square metre per mole (m 2/mol), but in practice, Of how strongly a chemical species attenuates light at a given wavelength. The molar extinction coefficient or molar attenuation coefficient is a measurement It isĭefined as the common logarithm of the ratio of incident to transmitted light power through a solution. Reduce storage space, and improve the accuracy with which working lower concentration solutions areĪbsorbance is a number that measures the attenuation of the transmitted light power in a solution. Stock solutions are used to save preparation Referred to as a working or final concentration, for actual use. Percent by volume (v/v) is the volume of solute divided by the total volume of the solution, multiplied by 100Ī stock solution is a concentrated solution that will be diluted to some lower concentration, which is often Percent by weight per volume (w/v), is the number of grams of solute in 100 mL of solution. Percent by mass (or weight), m/m or w/w is the mass of solute divided by the total mass of the solution, The percent concentration of a solution can be expressed as mass per mass (m/m or w/w), mass per volume (w/v), Mass concentration is the ratio of the mass of a solute to the volume of the solution, usually expressed in This contrasts with theĭefinition of molarity which is based on a specified volume of the solution. Molality is a measure of number of moles of solute present in 1 kg of solvent. 2.5 M (read as "2.5-molar") where "M" stands for mol/L. The value of molarity is also often expressed as "x-molar",Į.g. The most commonly used unit for molarity is the number of moles per liter, having the unit symbol mol/L or (see above), present in a certain volume of a solution. Molar concentration (also called molarity) is the number of molecules of a substance, expressed in mole units This amount canīe expressed as moles, mass units, or percents. ConcentrationĬoncentration of a solution is the amount of solute dissolved in a volume unit of the solution. Solute is a substance dissolved in another substance, known as a solvent. SolutionĪ solution, in chemistry, is a homogeneous mixture composed of two or more substances. SolventĪ solvent is a substance that dissolves a solute, resulting in a solution. SoluteĪ solute is a substance dissolved in another substance, known as a solvent. Unit or with a unit called the Dalton (Da). (MW or M.W., sometimes also called formula weight - FW or F.W.), albeit the latter is used either without any Substance is near identical to its molecular weight 
Molar mass is the mass of 1 mole of a substance, given in g/mol. 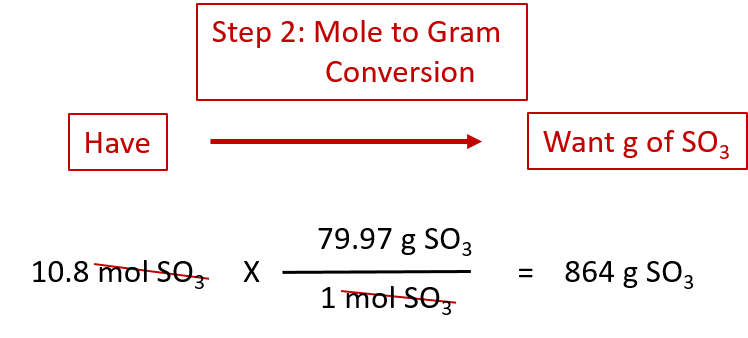
(the Avogadro constant) particles, which may be atoms, molecules, ions, or electrons. The mole is the unit of measurement for amount of substance. a pollution of 1 gram of benzene in a certain amount of water converts to N A/78.11≈ 7.Definitions and Formulas Definitions of Basic Terms Mole Using the above calculator you could find that e.g. Or 1 mole of a substance will contain Avogadro's number of that substance. The term " mole" is defined in that one mole of a substance with a molecular (or atomic) mass of one (1), will have a mass of 1 gram. It is defined to be 1/12 of the mass of one atom of carbon-12 and in older works is also abbreviated as "amu".Īlso, important in this field is Avogadro's number (N A) or Avogadro's constant (6.0221 x 10 23). In related terms, another unit of mass often used is Dalton (Da) or unified atomic mass unit (u) when describing atomic masses and molecular masses. Molecular mass or molar mass are used in stoichiometry calculations in chemistry. This Calculator has been tested on Internet Explorer version 6 only,įirefox might not show all fields correctly. For question or remarks please contact us. 
!!! Lenntech BV cannot be held responsible for errors in the calculation, Make sure you enter the molecule of crystallization at last (e.g. The calculator handles at most two different bracket levels. The molecular mass calculator will recognize the entered formula's, which are included in the list of organic compounds. Or you can choose by one of the next two option-lists, which contains a series of common organic compounds (including their chemical formula) and all the elements. This online calculator you can use for computing the average molecular weight (MW) of molecules by entering the chemical formulas (for example C3H4OH(COOH)3 ). Molecular Weight Calculator Molecular Weight Calculator Separation and Concentration Purification Request.Plant Inspection & Process Optimalisation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
